Core–Shell Structure and Interaction Mechanism of
γ-MnO2Coated Sulfur for Improved Lithium-Sulfur Batteries
Lubin Ni,a* Zhen Wu,aGangjin Zhao,aChunyu Sun,aChuanqiang Zhou,bXiangXiang Gong,band Guowang Diaoa*
aCollege of Chemistry and Chemical Engineering, Yangzhou University, Yangzhou 225002, Jiangsu, People’s Republic of China.
bTesting Center,Yangzhou University,Yangzhou, 225002 Jiangsu, People’s Republic of China.
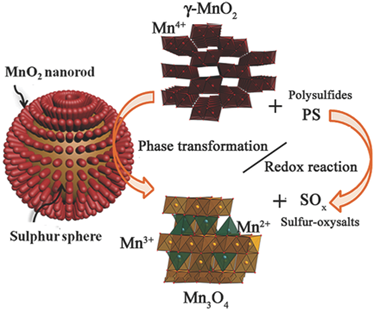
Abstract:Lithium-sulfur batteries have attracted worldwide interest due to their high theoreticalcapacity of 1672 mAh g−1 and low cost. However, the practical applications arehampered by capacity decay, mainly attributed to the polysulfide shuttle. Here, theauthors have fabricated a solid core–shellγ-MnO2-coated sulfur nanocompositethrough the redox reaction between KMnO4and MnSO4. The multifunctionalMnO2shell facilitates electron and Li+transport as well as efficiently preventspolysulfide dissolution via physical confinement and chemical interaction. Moreover,theγ-MnO2crystallographic form also provides one-dimensional (1D) tunnels forthe Li+incorporation to alleviate insoluble Li2S2/Li2S deposition at high dischargerate. More importantly, the MnO2phase transformation to Mn3O4occurs duringthe redox reaction between polysulfides andγ-MnO2is first thoroughly investigated.The S@γ-MnO2composite exhibits a good capacity retention of 82% after 300 cycles(0.5 C) and a fade rate of 0.07% per cycle over 600 cycles (1 C). The degradationmechanism can probably be elucidated that the decomposition of the surface Mn3O4phase is the cause of polysulfide dissolution. The recent work thus sheds new light onthe hitherto unknown surface interaction mechanism and the degradation mechanismof Li-S cells.
Small.2017, DOI:10.1002/smll.201603466.IF:8.315
文章链接:http://onlinelibrary.wiley.com/wol1/doi/10.1002/smll.201603466/full