Improved electrolysis of liquid ammonia for hydrogen generation via ammonium salt electrolyte and Pt/Rh/Ir electrocatalysts
Bao-Xia Dong*, Hui Tian, Yi-Chen Wu, Fan-Yan Bu, Wen-Long Liu,Yun-Lei Teng*, Guo-Wang Diao
School of Chemistry and Chemical Engineering, Yangzhou University
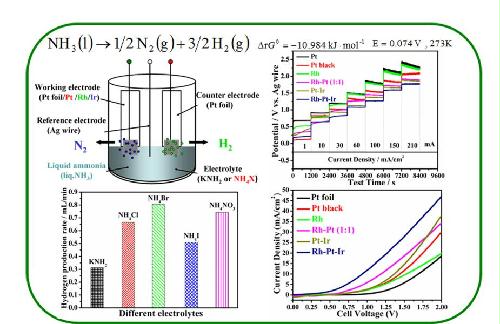
Abstract: Liquid ammonia electrolysis is conducted out at ambient temperature using different ammonium salt electrolytes with and without the reference electrode. It is determined that electrolytes containing NH4+increase the current density and reduce the solution resistance.Moreover, ammonium salts of NH4Br, NH4NO3and NH4Cl preserve >80% current efficiency and a 0.6-0.8 mL min-1average rate of H2generation during chronopotentiometry tests at 120 mA cm-2for 3 h. A minimum electrolysis voltage (Emin) of ca.1.2 V is required for the NH4Cl electrolyte when using Pt-foil as the anode and cathode. To decrease the overpotential of the anode reaction, five types of electrocatalysts were prepared and characterized: Pt-black, Rh, Pt-Ir, Rh-Pt and Ph-Pt-Ir alloys. Trimetallic Rh-Pt-Ir and bimetallic Pt-Ir, Rh-Pt (1:1) alloy electrodes exhibit a combined benefit of better activity and minimized deactivation. The ternary Rh-Pt-Ir alloy anode shows the best electrocatalytic activity with the lowest Eminof ca. 0.47 V and highest current density of 46.9 mA cm-2at 2.0 V. In comparison with the Pt-foil anode, the Emin is reduced by twothirds, and the current density is increased two-fold.
International journal of hydrogen energy, 2016, 41, 14507-14518
文章链接:http://www.sciencedirect.com/science/article/pii/S0360319916319346