Solvent- and Temperature-Induced Multiple Crystal Phases: Crystal Structure, Selective Adsorption, and Separation of Organic Dye in Three S‑Containing {[Cd(MIPA)]n}n− Homologues
Bao-Xia Dong,* Meng Tang, Wen-Long Liu,* Yi-Chen Wu, Yong-Mei Pan, Fan-Yan Bu, and Yun-Lei Teng
School of Chemistry and Chemical Engineering, Yangzhou University
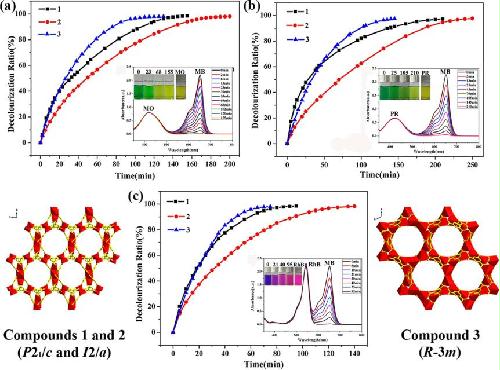
Abstract: Three S-containing highly porous metal−organic frameworks,namely, [((CH3)2NH2)Cd(MIPA)]n·xG (n = 4 (1), 2 (2), 1 (3) G = guest of DMF/DMA and H2O), featuring with high adsorption capacities and high selectivity for methylene blue (MB), were successfully assembled by subtly varying the synthesis conditions including solvents and temperatures. Compounds 1−3 crystallize in P21/c, I2/a, and R3̅m, respectively. The anionic frameworks of them are homologues and multiple crystal phases of the {[Cd(MIPA)]n}n−. Compounds 1 and 2 demonstrate the same 3,6-connected binodal net (42·6)(48·64·83), encapulating ca. 18 Å × 18 Å hexagonal channels and three other types of micropous channels. Compound 3 shows a different topology of a 4,4-connected binodal net (43·62·8), which has the highest crystalline density, highest solvent-accessible void, and accessible special surface area, as well as the highest symmetry among them. Circular channel, ca. 13 Å × 13 Å, and two other types of micropore channels are found in six directions. The dye adsorption process for these homologues is proven to be charge-selective and size-selective, in which only the cationic guest (MB+) with a suitable size could enter the frameworks of them and exchange with the dimethylammonium cations. The MB maximum adsorption capacities are 395.1 mg g−1 for 1, 286.6 mg g−1 for 2, and 373.2 mg g−1 for 3, respectively. Moreover, for three kinds of dye mixtures, 3 exhibits the best adsorption kinetics, which is probably due to the suitable pore size, highest solvent-accessible void, and the moderate maximum adsorption capacity of MB.
Crystal Growth&Design, 2016, 16(11), 6363-6370
文章链接:http://pubs.acs.org/doi/abs/10.1021/acs.cgd.6b00991