Structural, Magnetic, and Thermodynamic Evolutions of Zn-Doped Fe3O4Nanoparticles Synthesized Using a One-Step Solvothermal Method
Xin Liu,†Jun Liu,‡Shihui Zhang,†Zhaodong Nan,*,‡and Quan Shi*,†
†Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian 116023, China
‡College of Chemistry and Chemical Engineering, Yang Zhou University, Yangzhou 225002, China
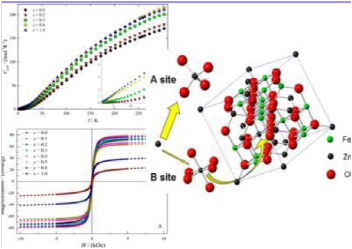
Abstract:Zn-doped Fe3O4magnetic nanoparticles represented as ZnxFe3‑xO4with different Zn contents of x varying from 0.0 to 1.0 were synthesized using a facile one-step solvothermal method. The Zn/Fe ratio in these particles could be accurately controlled using this facile synthesis technique. The ICP-OES and XRD measurements indicated that in the x range from 0 to 0.4 the doped Zn2+may replace the Fe3+at the A site and consequently the B-site Fe2+changed to Fe3+, while above 0.4 the Zn2+tends to replace the B-site Fe2+. The morphologies and size distributions of these samples characterized from the TEM showed that the nanoparticles appeared to aggregate into magnetic nanocrystal clusters with varying cluster sizes and different Zn doping contents. The magnetic measurement and Mössbauer spectra investigation revealed that the magnetic properties of the ZnxFe3‑xO4would exhibit a sensitive dependence with the doped Zn variations. Most importantly, the heat capacity studies illuminated that, at low temperatures, the samples could have a ferromagnetic contribution with x = 0.0 and 0.2 and turn to an antiferromagnetic contribution with x = 0.5, 0.8, and 1.0.
J. Phys. Chem. C 2016, 120, 1328−1341
文章链接:
JPCC 2016- 120- 1328